Have you ever eagerly awaited a delivery of battery strips for your latest project, only to find them rusting a few weeks later? It’s a frustrating scenario that plays out all too often in the world of battery building. Many enthusiasts and professionals alike have fallen into the trap of purchasing what they thought were high-quality connectors, only to discover they’ve been sold a cheaper alternative. The visual similarity between pure nickel and nickel-plated steel is striking, yet their performance could not be more different.
Choosing the wrong material is not just a matter of durability; it can significantly impact the safety and efficiency of your battery packs. Whether you are assembling a high-performance electric vehicle module, a custom e-bike battery, or even restringing a vintage guitar, understanding the nuances of these materials is crucial. This comprehensive guide will peel back the layers of confusion, providing you with the engineering insights and practical tests necessary to make an informed decision and avoid the hidden costs associated with “cheap” materials.
Table of Contents
Section 1: What Is the Difference Between Pure Nickel and Nickel Alloys?
At first glance, a strip of pure nickel and a strip of nickel-plated steel look virtually identical. They are both shiny, silver-colored metals that conduct electricity. However, beneath the surface, their atomic structures and physical properties diverge significantly. Understanding these differences is the first step in selecting the right material for your specific application.

Defining the Materials
Pure Nickel (N6/Ni200) is a material composed of at least 99.6% nickel. It is known for its excellent corrosion resistance and high electrical conductivity. Physically, it is a hard yet ductile metal that can be highly polished. A key characteristic that sets it apart in a workshop setting is its reaction to grinding; pure nickel typically produces very few sparks, and those that do appear are dim red in color.
Nickel-Plated Steel (NPS), on the other hand, is a composite material. It consists of a core made of steel (an alloy of iron and carbon) coated with a thin layer of nickel. This combination gives it a shiny appearance similar to pure nickel but with different mechanical properties. It tends to be “springy” and stiffer than pure nickel. When subjected to a grinding wheel, the steel core reveals itself by producing a shower of bright, branching yellow sparks, a telltale sign of iron content.
Why are both materials magnetic?
A common misconception among battery builders is that you can use a magnet to distinguish between pure nickel and nickel-plated steel. The logic often goes: “If it sticks to a magnet, it must be steel.” However, this is factually incorrect. Nickel is naturally a ferromagnetic metal, just like iron. Therefore, both pure nickel strips and nickel-plated steel strips will be attracted to a magnet.
While some specific nickel alloys might lose their magnetism due to the presence of other elements, the commercially pure nickel used in battery applications retains this property. So, while a magnet is a useful tool in a workshop, it is useless for telling these two specific materials apart. Relying on magnetism alone could lead you to mistakenly identify a high-quality pure nickel strip as a cheaper steel alternative.
Section 2: The Engineering Battle: Conductivity and Resistance
The primary function of these strips in a battery pack is to conduct electricity. Therefore, their electrical properties are of paramount importance. This is where the engineering reality separates the premium material from the budget option. The difference in resistance between pure nickel and steel is substantial and has real-world consequences for your battery’s performance.
Electrical Conductivity and Internal Resistance
Resistivity is a measure of how strongly a material opposes the flow of electric current. Pure nickel boasts a significantly lower electrical resistance compared to steel. Specifically, nickel-plated steel has an internal resistance that is approximately four times higher than that of pure nickel. This means that for the same dimensions, a pure nickel strip is a far more efficient conductor.
To put this into practical terms, let’s look at the current-carrying capacity. This is often measured in Amperes (A). Due to its lower resistance, a strip of pure nickel can handle a higher current density before overheating. A general rule of thumb for engineering calculations is:
- Pure Nickel: Can safely carry approximately 10A per mm² of cross-sectional area. For example, a standard 0.15mm x 7mm strip has a cross-section of 1.05mm² and can handle roughly 10.5A.
- Nickel-Plated Steel: Can only handle about 7A per mm². The same 0.15mm x 7mm strip would be limited to roughly 7.35A.
- Copper: For comparison, copper is an even better conductor, handling around 30A per mm², but it presents its own challenges in spot welding.
The Impact of Resistance on Battery Performance
Why does this resistance matter? It comes down to basic physics, specifically the formula $P = I^2R$, where $P$ is power lost as heat, $I$ is current, and $R$ is resistance. Since steel has higher resistance ($R$), pushing the same current ($I$) through it generates significantly more heat.
Heat Generation: Excess heat is the enemy of batteries. Heat generated in the connecting strips doesn’t just vanish; it radiates into the battery cells themselves. This can accelerate the degradation of the battery chemistry, shortening the lifespan of your expensive pack.
Voltage Drop: High resistance also causes “voltage sag.” When you demand power from your battery (like accelerating on an e-bike), some voltage is lost pushing current through the resistive strips. This results in less power reaching your motor, making the device feel sluggish. It effectively reduces the usable capacity of your battery, as the voltage will hit the cutoff point sooner under load.
Uneven Current Distribution: In large parallel groups, differences in resistance can lead to uneven current sharing. Some cells end up working harder than others, creating “hot spots” within the pack. These overworked cells fail earlier, dragging the performance of the entire battery down with them.
| Feature | Pure Nickel (Ni200) | Nickel-Plated Steel |
|---|---|---|
| Resistance | Low (~7-10 µΩ/cm) | High (~4x Pure Nickel) |
| Conductivity | High (~10A/mm²) | Low (~7A/mm²) |
| Heat Generation | Low | High |
| Voltage Sag | Minimal | Significant under load |
Section 3: Durability and Environmental Resistance
Beyond electrical performance, the physical durability of your connection material determines how long your battery pack will survive in the real world. Environmental factors like humidity, salt, and temperature fluctuations constantly attack metal components. This is where the protective qualities of your chosen strip become critical.

Corrosion Resistance: The Salt Spray Reality
Pure nickel is naturally resistant to oxidation and corrosion. It creates a passive oxide layer that protects it from the elements. This makes it an ideal choice for applications in humid climates or marine environments where salt air is a constant threat.
Nickel-plated steel, however, has a significant weakness: its “Achilles Heel.” The corrosion resistance relies entirely on the thin layer of nickel plating. The moment this plating is scratched, cut, or damaged—which inevitably happens during the assembly process—the underlying steel core is exposed. Once exposed to moisture or air, the steel oxidizes rapidly.
A simple yet effective demonstration of this is the “Saltwater Test.” If you scratch both materials and submerge them in saltwater for 24 hours, the steel core of the plated strip will begin to rust visibly, while the pure nickel strip will remain unaffected. This rust not only weakens the structural integrity of the tab but also increases electrical resistance further, creating a vicious cycle of heating and degradation.
Environmental Impact and Sustainability
The choice of material also has broader environmental implications. Using pure nickel extends the operational lifespan of a battery pack. A longer-lasting battery means fewer replacements are needed over the life of the device, directly reducing electronic waste.
Battery disposal is a significant environmental challenge due to the toxic chemicals and heavy metals involved. Premature battery failure caused by corroded or overheated low-quality connectors adds unnecessary volume to waste streams. Manufacturers who choose pure nickel are not just making a technical decision; they are making a sustainability choice. By aligning with “green” manufacturing principles and ensuring durability, builders contribute to a reduction in resource consumption and waste generation.
Section 4: The Spot Welding Dilemma
If pure nickel is superior in performance and durability, why is nickel-plated steel so prevalent? One major reason, aside from cost, is the ease of manufacturing. The spot welding process—the standard method for connecting strips to battery cells—behaves very differently depending on the material you are using.

Why is Steel Easier to Weld?
Resistance spot welding works by passing a high current through the materials to be joined. The resistance of the material to this current flow generates heat, which melts the metal at the contact point and creates a weld nugget. Herein lies the paradox: High resistance generates heat.
Because steel has high electrical resistance, it heats up very quickly when current is applied. This makes it incredibly easy to weld. You can achieve strong bonds with relatively inexpensive, low-power spot welders, even simple setups powered by a 12V car battery.
Pure nickel, with its low resistance, allows the current to pass through easily without generating as much heat. To get a good weld with pure nickel, you need significantly more energy. This requires high-power, industrial-grade spot welders capable of delivering precise, high-current pulses (such as the Kweld or Glitter 811H). Builders using underpowered equipment often struggle to get pure nickel to stick, leading to weak welds and frustration.
Advanced Techniques: The “Sandwich” Method
For applications requiring extreme current, even pure nickel might have too much resistance. In these cases, builders turn to copper. However, copper is even harder to weld than nickel due to its extreme conductivity. A clever workaround discussed by experts is the “Sandwich” method.
This involves placing a highly conductive copper strip on the battery terminal and placing a layer of nickel or stainless steel on top of it. The welder probes press onto the top layer. The higher resistance of the top layer generates the necessary heat, which then melts through to the copper and the battery terminal, creating a bond. Some users have found that 304 stainless steel makes an excellent “cap” for this technique, providing superior weld strength.
Common Welding Problems and Solutions
Achieving the perfect weld requires balancing power and timing:
- Burnt Welds: Often caused by too much pulse time on nickel strips. The excess energy burns the metal rather than melting it cleanly.
- Weak Welds: Usually the result of insufficient power when working with pure nickel. The current passes through without generating enough heat to fuse the metals.
- Corrosion at the Weld: With nickel-plated steel, the welding process itself can damage the plating. The heat-affected zone around the weld can become a prime spot for rust to start, compromising the connection over time.
Section 5: Application Guide: When to Use Which?
Not every battery pack needs the premium performance of pure nickel. Engineering is about choosing the right tool for the job. Here is a breakdown of when to invest in pure nickel and when plated steel might be sufficient.

When to Choose Pure Nickel
Pure nickel is the mandatory choice for high-drain and mission-critical applications. If your device pulls significant current or operates in harsh conditions, do not compromise.
- High-Drain Devices: Electric vehicles (EVs), e-bikes, electric skateboards, power tools, and racing drones.
- Critical Environments: Medical devices where failure is not an option, aerospace applications, and marine equipment exposed to salt and humidity.
- Long-Term Reliability: Solar energy storage banks and UPS systems that are expected to operate for years without maintenance.
When to Choose Nickel Plated Steel
Nickel-plated steel has its place in the market, primarily where cost is the main driver and performance demands are low.
- Low-Drain Devices: Flashlights, USB power banks, and children’s toys where the current draw is minimal.
- Cost-Sensitive Projects: Budget builds where saving every cent is critical.
- Prototyping: Temporary packs built for testing fit and function, where long-term durability is not a concern.
A Side Note: Guitar Strings (The Tonal Difference)
Interestingly, the debate between pure nickel and plated steel isn’t limited to batteries. Musicians have long argued over the merits of each for guitar strings. Vintage enthusiasts prefer pure nickel strings for their warm, mellow, “broken-in” tone. In contrast, nickel-plated steel strings are brighter, louder, and “twangier” due to the higher iron content affecting the magnetic pickup field. While plated strings are harder and may last longer against fret wear, many players find their initial sound too harsh. This parallel highlights how the fundamental physical properties of these materials—magnetic response and hardness—manifest differently across various industries.
Section 6: How to Test and Identify Your Material
With the market flooded with mislabeled products, knowing how to verify what you bought is an essential skill. You cannot rely on visual inspection alone. Here are three proven methods to test your strips.

The Spark Test (Grinding)
This is the most immediate and dramatic test. Using a Dremel tool or a bench grinder:
- Touch the spinning wheel to the metal strip.
- Observe the sparks:
- Steel: You will see a shower of bright, yellow sparks that branch out (like a sparkler). This is characteristic of burning carbon/iron.
- Pure Nickel: You will see very few sparks, if any. The sparks will be short, dim, and dark red in color.
The Salt Water Corrosion Test
This test simulates accelerated aging.
- Use sandpaper or a file to deeply scratch the surface of the strip, ensuring you cut through any potential plating.
- Submerge the strip in a glass of strong saltwater solution.
- Wait 24 hours.
- Results: If you see rust forming on the scratches, it is steel. If the strip remains clean and shiny, it is pure nickel.
The Resistance Test
For a quantitative approach, you can measure the resistance directly using a high-precision 4-wire ohm meter (like the YR1035+).
- Measure a specific length of the strip (e.g., 10cm).
- Compare the reading to known standards. Real-world tests often show a 10cm strip of pure nickel measuring around 9.8 milliohms, while a steel strip of the same size measures around 14.8 milliohms.
- While the theoretical difference is 4x, real-world measurements often show a ~50% difference due to plating thickness and alloy variations, but this is still distinct enough to identify the material.
Section 7: Deep Dive into Plating Technologies (Electroless vs. Electrolytic)
If you do decide to use plated materials, or encounter them in industrial settings, it’s worth understanding how the plating is applied. Not all plating is created equal.
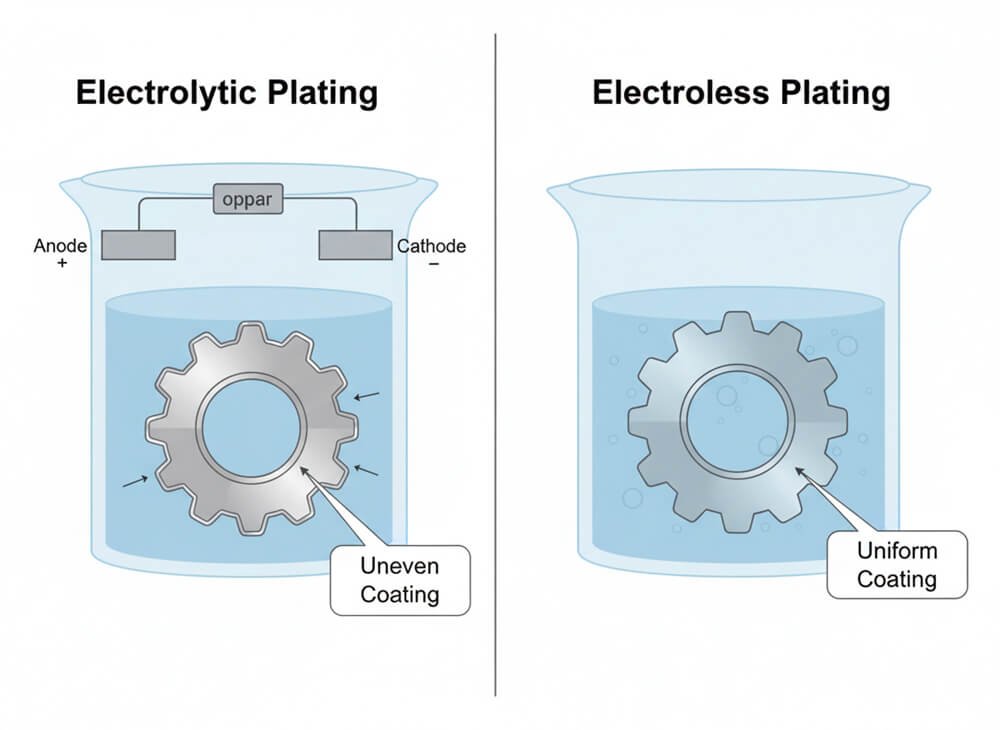
Electrolytic Nickel Plating
This is the standard method used for most consumer goods. It involves using an electric current to deposit nickel onto a conductive surface. While it is cost-effective and produces a bright, shiny finish, it has a major drawback: the “dog-bone” effect. The electrical current density is higher at edges and corners, leading to thicker plating in those areas and thinner plating in recesses. This lack of uniformity can leave parts of the steel core vulnerable.
Electroless Nickel Plating
This method relies on a chemical autocatalytic reaction rather than electricity. It deposits a nickel-phosphorus alloy evenly across the entire surface of the part, regardless of its shape. This results in a perfectly uniform coating, even in holes and threads. Electroless plating offers superior hardness and corrosion resistance, making it ideal for complex industrial parts like valves and hard drive components. While more expensive, it provides a much higher level of protection.
Which Plating is Used for Battery Strips?
The vast majority of budget “nickel strips” on the market utilize Electrolytic Plating. It is cheaper and faster to produce continuous rolls this way. However, this means the protection is often uneven, and the cut edges of the strip are completely unplated, leaving the steel core exposed to the air from day one.
FAQ
No, you cannot. Both pure nickel and steel are ferromagnetic materials, meaning they are both attracted to magnets. A magnet test will give you a “false positive” for steel every time.
Yes. The nickel plating is a protective barrier, but once it is scratched, cut, or spot-welded, the underlying steel is exposed. In humid environments, this exposed steel will rust rapidly.
Steel is generally easier to weld with lower-power equipment because its high resistance generates heat quickly. Pure nickel requires higher power settings or industrial-grade welders to achieve a secure bond due to its low resistance.
A pure nickel strip of 0.15mm x 7mm can handle approximately 10-11 Amps continuously. A nickel-plated steel strip of the same dimensions is generally limited to about 7-7.5 Amps.
Yes, pure nickel is a commodity metal that is significantly more expensive than steel. You can expect to pay roughly 3 to 4 times more for pure nickel strips compared to plated steel.
Conclusion
The choice between pure nickel and nickel-plated steel is a trade-off between performance and longevity versus cost and ease of use. For any serious battery project—especially those involving transportation like e-bikes or EVs—pure nickel is the only safe engineering choice. The lower resistance, heat management, and corrosion protection it offers are essential for safety.
While plated steel has its place in low-cost, low-power electronics, using it in high-drain applications is a risk that outweighs the savings. We highly recommend verifying your materials using the spark or saltwater tests to ensure you are getting what you paid for.
We understand that quality components are the foundation of a reliable battery pack. We offer certified, high-purity nickel strips designed to meet the rigorous demands of modern battery engineering. Don’t let a cheap connector be the weak link in your system.


